API intermediates from Intermediate
Manufacturer are substances or agents that are utilized as
raw materials in the production of active pharmaceutical ingredients (API).
These intermediates are produced via amination, hydrogenation, alkoxylation,
hydroformylation, electrochemistry, acetylene chemistry, phosgene chemistry,
and other technologies.
The API intermediate industry is being driven by the
increased acceptance of generic medications, and specialty medicine, a spike in
demand for biopharmaceuticals, and a growth in abbreviated new drug
applications. Furthermore, an increase in illness incidence and an increase in
the elderly population prone to different diseases and disorders are expected
to drive market expansion. R&D initiatives for the creation and enhancement
of medications for chronic illnesses, as well as FDA approvals for novel compounds,
give fresh market expansion potential.
The API intermediate market is divided into four
sections: product, intermediate type, therapeutic use, and geography. It is
divided into chemical intermediates and biotech intermediates based on the
product.
The pharmaceutical sector is developing and renewing
itself all the time. As the need for pharmaceutical intermediates from Intermediate Supplier China grows, a lot of pharmaceutical businesses
and industry participants are adjusting to new sales methods.
What precisely
are pharmacological intermediates?
Medicinal intermediates are fine chemicals that are
created as intermediary compounds during the manufacturing of active
pharmaceutical components (APIs). Intermediates are byproducts of the reactions
that occur throughout the API manufacturing process. Depending on the analysis,
each reaction in the manufacturing process may yield many intermediates that
can be employed as precursors for additional active pharmaceutical components.
Further refining methods can be used to convert intermediates to active
components.
There are several variables and studies that separate active components and molecules from pharmaceutical intermediates. The final products of raw materials are active ingredients and specialty chemicals, whereas chemical intermediates from Intermediate Manufacturer China are byproducts of the API production process. Furthermore, unlike active substances that include particular compounds, intermediates are not subject to approval or regulation.
As global pharmaceutical activity grows, new inventions emerge and the need for pharmaceutical intermediates producers rises. The worldwide pharmaceuticals and packaging industry has expanded as a result of pharmaceutical businesses implementing regulated pharmaceutical operations.
Follow us on Facebook

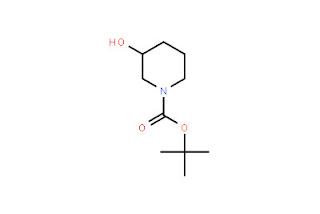
Piperidine.jpg)


Benzene.jpg)

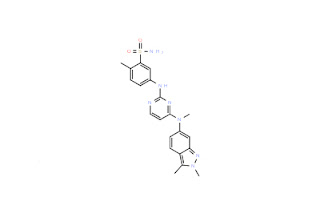

![5-(4-Phenoxyphenyl)-7H-Pyrrolo[2,3-D]PyriMidin-4-YlaMine Intermediate Supplier China](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEhFOc8oJEoM1fWnj6Xu9lkpZDtmzsrYChnTvlLLidJCynmyTXuzDGuqxjtfVWaac7FzP9uLeswnxxRP3ZIMm55jaHd5lXH3GoxrMqKN3RXrekBgABbs0UiXBtWS8YqBjO4ABqYxbKvGFf2jzSskD0BCylKWnEtAH7f-N2BUZUZC_kkhda3WLrS1WmVE/w320-h237/5-(4-Phenoxyphenyl)-7H-Pyrrolo%5B2,3-D%5DPyriMidin-4-YlaMine.jpg)
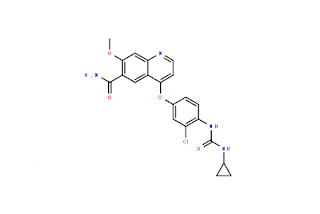
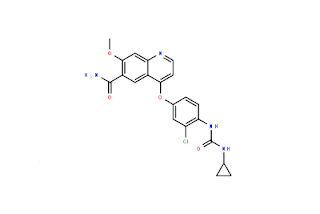
-N4-(3-Chloro-4-Fluorophenyl)-7-((Tetrahydrofuran-3-Yl)Oxy)Quinazoline-4,6-Diamine.jpg)
